 Chemistry - Periodic Table 2 Chemistry - Periodic Table 2

The 118 elements are arranged in a particular order. Chemical shorts are used for reasons of space, the names of the elements are omitted here. Incidentally, these does not base on the English language (iron -
ferrum) but the Latin.
The atomic numbers are striking, which specify an order. That are the numbers of charges, the protons in the atomic nucleus and the electrons in the atomic shell.
The number of neutrons is not yet considered here.
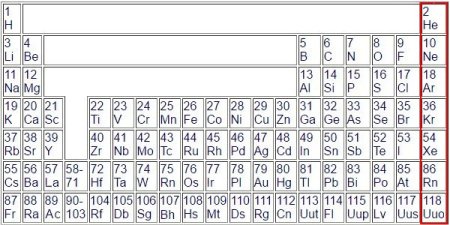
The gap at the top in the middle is due to the fact that the elements of the eight main groups are to be combined, 1 and 2 to the left and 3 to 8 to the right. In the
middle the side groups, incidentally all metals. On the lower left of the side groups there are placeholders. The atomic numbers from 58 to 71 are for the isotopes of Lanthan (Lanthanides) and from 90 to
103 for the isotopes of Actinium (Actinides).
Its name has the system from the periods, these are the lines. In the chapter Electrons, you learned that there are main quantum numbers for differentiating electron
groups from 1 to 7. Thus, with the two elements hydrogen and helium, there is only one main group in the not fully occupied first period. This increases up to the
seven major groups present in the seventh line (period).
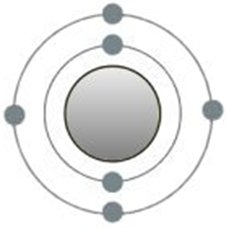
The electrons collected in the last major quantum number are all in the outer side of the atomic shell, also called 'valence electrons'. Their numbers are very
important for reactions with other atoms, since the only way to exchange electrons is here, but only if the maximum possible number is not reached. The number of
valence electrons determines the belonging to a particular main group, with the exception of helium.
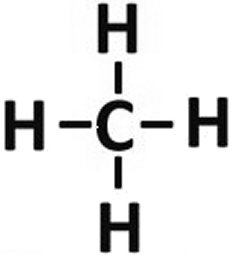
The carbon atom lacks four valence electrons, which is why it can, for example, bind four hydrogen atoms with only one electron on the outermost shell. Carbon is therefore referred to as tetravalent and hydrogen as
monovalent. Of the oxygen atoms, there are only two on the carbon, as the name carbondioxide already suggests.
The formation of such a molecule is usually the result of a reaction. It is said that carbon and oxygen react with each other, which in a reaction equation for the formation of water looks like this, for example:
2 H2 + O2 -> 2 H2O
Why there is the '2' before the 'H' and the ' H2O' respectively? Because you always need two atoms of hydrogen and only one atom of oxygen, which occurs naturally as a molecule of two atoms. So you create
two molecules, which becomes clear on the right-hand side of the reaction equation. This quantity then comes before the complete name of the molecule.
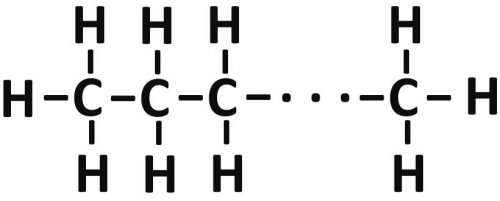
Graphically, the respective first letters of the Latin name for the hydrocarbon are created here, with the lines in between for the number of bonds. The lines can also be multiple and the carbon can also combine with itself,
thereby forming a chain, such as occurs in diesel fuel, for example.
| It is common in chemistry that certain compounds are abandoned because of other, more desirable
ones. |
|